You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000013_04120
You are here: Home > Sequence: MGYG000000013_04120
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Bacteroides sp902362375 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; Bacteroides sp902362375 | |||||||||||
| CAZyme ID | MGYG000000013_04120 | |||||||||||
| CAZy Family | GH20 | |||||||||||
| CAZyme Description | Chitobiase | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 210188; End: 212713 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH20 | 319 | 731 | 5.5e-97 | 0.9554896142433235 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd06569 | GH20_Sm-chitobiase-like | 0.0 | 314 | 741 | 1 | 427 | The chitobiase of Serratia marcescens is a beta-N-1,4-acetylhexosaminidase with a glycosyl hydrolase family 20 (GH20) domain that hydrolyzes the beta-1,4-glycosidic linkages in oligomers derived from chitin. Chitin is degraded by a two step process: i) a chitinase hydrolyzes the chitin to oligosaccharides and disaccharides such as di-N-acetyl-D-glucosamine and chitobiose, ii) chitobiase then further degrades these oligomers into monomers. The GH20 hexosaminidases are thought to act via a catalytic mechanism in which the catalytic nucleophile is not provided by solvent or the enzyme, but by the substrate itself. |
| cd06563 | GH20_chitobiase-like | 8.57e-106 | 319 | 732 | 2 | 345 | The chitobiase of Serratia marcescens is a beta-N-1,4-acetylhexosaminidase with a glycosyl hydrolase family 20 (GH20) domain that hydrolyzes the beta-1,4-glycosidic linkages in oligomers derived from chitin. Chitin is degraded by a two step process: i) a chitinase hydrolyzes the chitin to oligosaccharides and disaccharides such as di-N-acetyl-D-glucosamine and chitobiose, ii) chitobiase then further degrades these oligomers into monomers. This GH20 domain family includes an N-acetylglucosamidase (GlcNAcase A) from Pseudoalteromonas piscicida and an N-acetylhexosaminidase (SpHex) from Streptomyces plicatus. SpHex lacks the C-terminal PKD (polycystic kidney disease I)-like domain found in the chitobiases. The GH20 hexosaminidases are thought to act via a catalytic mechanism in which the catalytic nucleophile is not provided by solvent or the enzyme, but by the substrate itself. |
| pfam00728 | Glyco_hydro_20 | 7.25e-105 | 319 | 731 | 2 | 345 | Glycosyl hydrolase family 20, catalytic domain. This domain has a TIM barrel fold. |
| COG3525 | Chb | 3.81e-92 | 265 | 801 | 206 | 699 | N-acetyl-beta-hexosaminidase [Carbohydrate transport and metabolism]. |
| cd06562 | GH20_HexA_HexB-like | 3.94e-60 | 319 | 730 | 2 | 323 | Beta-N-acetylhexosaminidases catalyze the removal of beta-1,4-linked N-acetyl-D-hexosamine residues from the non-reducing ends of N-acetyl-beta-D-hexosaminides including N-acetylglucosides and N-acetylgalactosides. The hexA and hexB genes encode the alpha- and beta-subunits of the two major beta-N-acetylhexosaminidase isoenzymes, N-acetyl-beta-D-hexosaminidase A (HexA) and beta-N-acetylhexosaminidase B (HexB). Both the alpha and the beta catalytic subunits have a TIM-barrel fold and belong to the glycosyl hydrolase family 20 (GH20). The HexA enzyme is a heterodimer containing one alpha and one beta subunit while the HexB enzyme is a homodimer containing two beta-subunits. Hexosaminidase mutations cause an inability to properly hydrolyze certain sphingolipids which accumulate in lysosomes within the brain, resulting in the lipid storage disorders Tay-Sachs and Sandhoff. Mutations in the alpha subunit cause in a deficiency in the HexA enzyme and result in Tay-Sachs, mutations in the beta-subunit cause in a deficiency in both HexA and HexB enzymes and result in Sandhoff disease. In both disorders GM(2) gangliosides accumulate in lysosomes. The GH20 hexosaminidases are thought to act via a catalytic mechanism in which the catalytic nucleophile is not provided by solvent or the enzyme, but by the substrate itself. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QNL39428.1 | 0.0 | 1 | 841 | 1 | 841 |
| QUT79935.1 | 0.0 | 1 | 841 | 1 | 841 |
| QDM09738.1 | 0.0 | 1 | 841 | 1 | 841 |
| QUT31033.1 | 0.0 | 1 | 841 | 1 | 841 |
| QUR46113.1 | 0.0 | 1 | 841 | 1 | 841 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 1QBA_A | 6.45e-95 | 50 | 803 | 44 | 823 | BACTERIALCHITOBIASE, GLYCOSYL HYDROLASE FAMILY 20 [Serratia marcescens],1QBB_A BACTERIAL CHITOBIASE COMPLEXED WITH CHITOBIOSE (DINAG) [Serratia marcescens] |
| 1C7T_A | 1.24e-94 | 50 | 803 | 44 | 823 | ChainA, BETA-N-ACETYLHEXOSAMINIDASE [Serratia marcescens] |
| 1C7S_A | 6.42e-94 | 50 | 803 | 44 | 823 | ChainA, BETA-N-ACETYLHEXOSAMINIDASE [Serratia marcescens] |
| 6EZR_A | 1.82e-63 | 193 | 771 | 142 | 638 | Crystalstructure of GH20 Exo beta-N-Acetylglucosaminidase from Vibrio harveyi [Vibrio harveyi],6EZR_B Crystal structure of GH20 Exo beta-N-Acetylglucosaminidase from Vibrio harveyi [Vibrio harveyi],6EZS_A Crystal structure of GH20 Exo beta-N-Acetylglucosaminidase from Vibrio harveyi in complex with N-acetylglucosamine [Vibrio harveyi],6EZS_B Crystal structure of GH20 Exo beta-N-Acetylglucosaminidase from Vibrio harveyi in complex with N-acetylglucosamine [Vibrio harveyi],6K35_A Crystal structure of GH20 exo beta-N-acetylglucosaminidase from Vibrio harveyi in complex with NAG-thiazoline [Vibrio harveyi],6K35_B Crystal structure of GH20 exo beta-N-acetylglucosaminidase from Vibrio harveyi in complex with NAG-thiazoline [Vibrio harveyi] |
| 6EZT_A | 2.17e-62 | 193 | 771 | 139 | 635 | Crystalstructure of GH20 Exo beta-N-Acetylglucosaminidase D437A inactive mutant from Vibrio harveyi [Vibrio harveyi],6EZT_B Crystal structure of GH20 Exo beta-N-Acetylglucosaminidase D437A inactive mutant from Vibrio harveyi [Vibrio harveyi] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| P13670 | 5.79e-95 | 47 | 801 | 66 | 846 | N,N'-diacetylchitobiase OS=Vibrio harveyi OX=669 GN=chb PE=1 SV=1 |
| Q54468 | 8.29e-94 | 50 | 803 | 71 | 850 | Chitobiase OS=Serratia marcescens OX=615 GN=chb PE=1 SV=1 |
| Q04786 | 2.79e-91 | 52 | 801 | 47 | 809 | Beta-hexosaminidase OS=Vibrio vulnificus OX=672 GN=hex PE=3 SV=1 |
| P49007 | 1.15e-85 | 162 | 753 | 183 | 750 | Beta-hexosaminidase B OS=Pseudoalteromonas piscicida OX=43662 GN=nag096 PE=3 SV=1 |
| P96155 | 1.01e-60 | 187 | 725 | 135 | 601 | Beta-hexosaminidase OS=Vibrio furnissii OX=29494 GN=exoI PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as LIPO
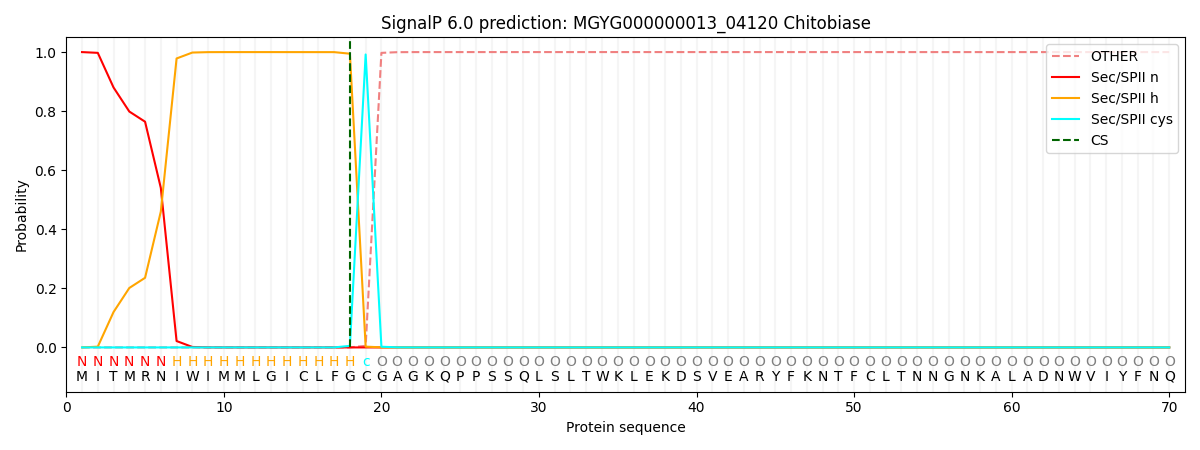
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000000 | 0.000015 | 1.000028 | 0.000000 | 0.000000 | 0.000000 |
