You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000117_00149
You are here: Home > Sequence: MGYG000000117_00149
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Barnesiella sp003150885 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Barnesiellaceae; Barnesiella; Barnesiella sp003150885 | |||||||||||
| CAZyme ID | MGYG000000117_00149 | |||||||||||
| CAZy Family | GH16 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 147902; End: 149356 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH16 | 141 | 388 | 5.8e-34 | 0.9911504424778761 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd08023 | GH16_laminarinase_like | 1.23e-21 | 138 | 388 | 1 | 235 | Laminarinase, member of the glycosyl hydrolase family 16. Laminarinase, also known as glucan endo-1,3-beta-D-glucosidase, is a glycosyl hydrolase family 16 member that hydrolyzes 1,3-beta-D-glucosidic linkages in 1,3-beta-D-glucans such as laminarins, curdlans, paramylons, and pachymans, with very limited action on mixed-link (1,3-1,4-)-beta-D-glucans. |
| cd00413 | Glyco_hydrolase_16 | 7.80e-18 | 140 | 388 | 1 | 210 | glycosyl hydrolase family 16. The O-Glycosyl hydrolases are a widespread group of enzymes that hydrolyse the glycosidic bond between two or more carbohydrates, or between a carbohydrate and a non-carbohydrate moiety. A glycosyl hydrolase classification system based on sequence similarity has led to the definition of more than 95 different families inlcuding glycosyl hydrolase family 16. Family 16 includes lichenase, xyloglucan endotransglycosylase (XET), beta-agarase, kappa-carrageenase, endo-beta-1,3-glucanase, endo-beta-1,3-1,4-glucanase, and endo-beta-galactosidase, all of which have a conserved jelly roll fold with a deep active site channel harboring the catalytic residues. |
| TIGR04183 | Por_Secre_tail | 2.94e-09 | 408 | 484 | 1 | 72 | Por secretion system C-terminal sorting domain. Species that include Porphyromonas gingivalis, Fibrobacter succinogenes, Flavobacterium johnsoniae, Cytophaga hutchinsonii, Gramella forsetii, Prevotella intermedia, and Salinibacter ruber average twenty or more copies of a C-terminal domain, represented by this model, associated with sorting to the outer membrane and covalent modification. |
| cd02178 | GH16_beta_agarase | 1.45e-08 | 185 | 388 | 61 | 257 | Beta-agarase, member of glycosyl hydrolase family 16. Beta-agarase is a glycosyl hydrolase family 16 (GH16) member that hydrolyzes the internal beta-1,4-linkage of agarose, a hydrophilic polysaccharide found in the cell wall of Rhodophyceaea, marine red algae. Agarose is a linear chain of galactose units linked by alternating L-alpha-1,3- and D-beta-1,4-linkages that are additionally modified by a 3,6-anhydro-bridge. Agarose forms thermo-reversible gels that are widely used in the food industry or as a laboratory medium. While beta-agarases are also found in two other families derived from the sequence-based classification of glycosyl hydrolases (GH50, and GH86) the GH16 members are most abundant. This domain adopts a curved beta-sandwich conformation, with a tunnel-shaped active site cavity, referred to as a jellyroll fold. |
| cd02177 | GH16_kappa_carrageenase | 4.84e-08 | 222 | 388 | 94 | 268 | Kappa-carrageenase, member of glycosyl hydrolase family 16. Kappa-carrageenase is a glycosyl hydrolase family 16 (GH16) member that hydrolyzes the internal beta-1,4-linkage of kappa-carrageenans, a hydrophilic polysaccharide found in the cell wall of Rhodophyceaea, marine red algae. Carrageenans are linear chains of galactose units linked by alternating D-alpha-1,3- and D-beta-1,4-linkages that are additionally modified by a 3,6-anhydro-bridge. Depending on the position and number of sulfate ester modifications they are subdivided into kappa-, iota-, and lambda-carrageenases, kappa being modified once. Carrageenans form thermo-reversible gels widely used for industrial applications. Kappa-carrageenases exist in bacteria belonging to at least three phylogenetically distant branches, including pseudoalteromonas, planctomycetes, and baceroidetes. This domain adopts a curved beta-sandwich conformation, with a tunnel-shaped active site cavity, referred to as a jellyroll fold. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QVL33586.1 | 5.96e-150 | 23 | 389 | 217 | 587 |
| QEH37426.1 | 3.49e-140 | 23 | 388 | 205 | 574 |
| QUE50368.1 | 7.58e-105 | 23 | 388 | 161 | 519 |
| QGT72065.1 | 4.26e-92 | 23 | 390 | 46 | 415 |
| AHF11964.1 | 2.56e-91 | 23 | 394 | 52 | 426 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 2VY0_A | 7.39e-11 | 187 | 389 | 68 | 260 | TheX-ray structure of endo-beta-1,3-glucanase from Pyrococcus furiosus [Pyrococcus furiosus],2VY0_B The X-ray structure of endo-beta-1,3-glucanase from Pyrococcus furiosus [Pyrococcus furiosus] |
| 5OCR_A | 5.78e-08 | 222 | 391 | 101 | 286 | Crystalstructure of the kappa-carrageenase zobellia_236 from Zobellia galactanivorans [Zobellia galactanivorans],5OCR_B Crystal structure of the kappa-carrageenase zobellia_236 from Zobellia galactanivorans [Zobellia galactanivorans],5OCR_C Crystal structure of the kappa-carrageenase zobellia_236 from Zobellia galactanivorans [Zobellia galactanivorans],5OCR_D Crystal structure of the kappa-carrageenase zobellia_236 from Zobellia galactanivorans [Zobellia galactanivorans] |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as SP
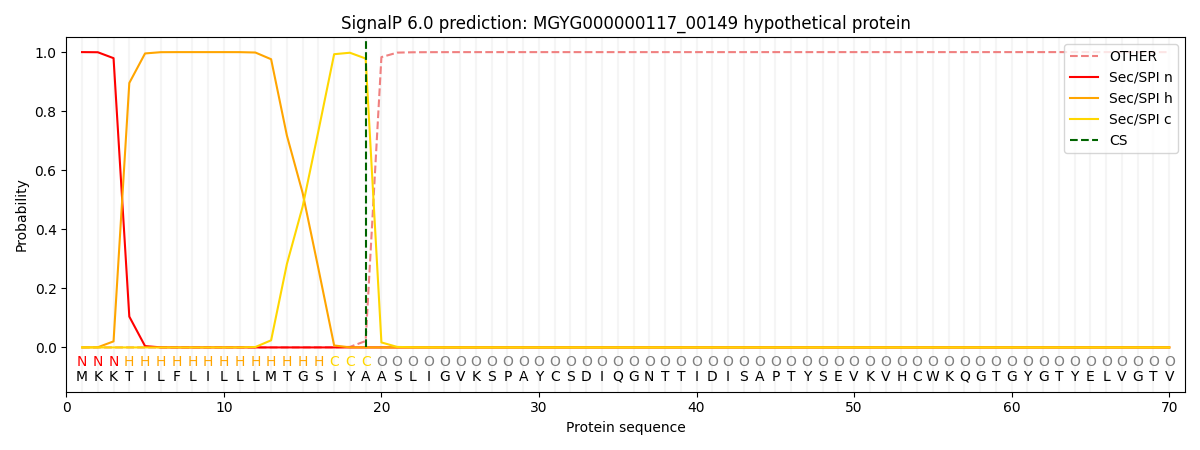
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000326 | 0.998994 | 0.000177 | 0.000170 | 0.000154 | 0.000148 |

