You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000196_01712
You are here: Home > Sequence: MGYG000000196_01712
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Bacteroides thetaiotaomicron | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; Bacteroides thetaiotaomicron | |||||||||||
| CAZyme ID | MGYG000000196_01712 | |||||||||||
| CAZy Family | GH38 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 160923; End: 163472 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH38 | 12 | 266 | 7.7e-33 | 0.9256505576208178 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG0383 | AMS1 | 7.28e-19 | 105 | 814 | 286 | 917 | Alpha-mannosidase [Carbohydrate transport and metabolism]. |
| PRK09819 | PRK09819 | 3.63e-16 | 400 | 828 | 388 | 861 | mannosylglycerate hydrolase. |
| pfam01074 | Glyco_hydro_38 | 6.83e-13 | 12 | 189 | 2 | 172 | Glycosyl hydrolases family 38 N-terminal domain. Glycosyl hydrolases are key enzymes of carbohydrate metabolism. |
| cd10789 | GH38N_AMII_ER_cytosolic | 5.62e-11 | 46 | 189 | 32 | 171 | N-terminal catalytic domain of endoplasmic reticulum(ER)/cytosolic class II alpha-mannosidases; glycoside hydrolase family 38 (GH38). The subfamily is represented by Saccharomyces cerevisiae vacuolar alpha-mannosidase Ams1, rat ER/cytosolic alpha-mannosidase Man2C1, and similar proteins. Members in this family share high sequence similarity. None of them have any classical signal sequence or membrane spanning domains, which are typical of sorting or targeting signals. Ams1 functions as a second resident vacuolar hydrolase in S. cerevisiae. It aids in recycling macromolecular components of the cell through hydrolysis of terminal, non-reducing alpha-d-mannose residues. Ams1 utilizes both the cytoplasm to vacuole targeting (Cvt, nutrient-rich conditions) and autophagic (starvation conditions) pathways for biosynthetic delivery to the vacuole. Man2C1is involved in oligosaccharide catabolism in both the ER and cytosol. It can catalyze the cobalt-dependent cleavage of alpha 1,2-, alpha 1,3-, and alpha 1,6-linked mannose residues. Members in this family are retaining glycosyl hydrolases of family GH38 that employs a two-step mechanism involving the formation of a covalent glycosyl-enzyme complex. Two carboxylic acids positioned within the active site act in concert: one as a catalytic nucleophile and the other as a general acid/base catalyst. |
| smart00872 | Alpha-mann_mid | 7.48e-09 | 297 | 344 | 14 | 62 | Alpha mannosidase, middle domain. Members of this entry belong to the glycosyl hydrolase family 38, This domain, which is found in the central region adopts a structure consisting of three alpha helices, in an immunoglobulin/albumin-binding domain-like fold. The domain is predominantly found in the enzyme alpha-mannosidase. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QUT69752.1 | 0.0 | 1 | 849 | 1 | 849 |
| AAO79177.1 | 0.0 | 1 | 849 | 1 | 849 |
| QMW86426.1 | 0.0 | 1 | 849 | 1 | 849 |
| ALJ43681.1 | 0.0 | 1 | 849 | 1 | 849 |
| QQA07855.1 | 0.0 | 1 | 849 | 1 | 849 |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| Q54K67 | 2.60e-10 | 75 | 844 | 315 | 1082 | Alpha-mannosidase G OS=Dictyostelium discoideum OX=44689 GN=manG PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
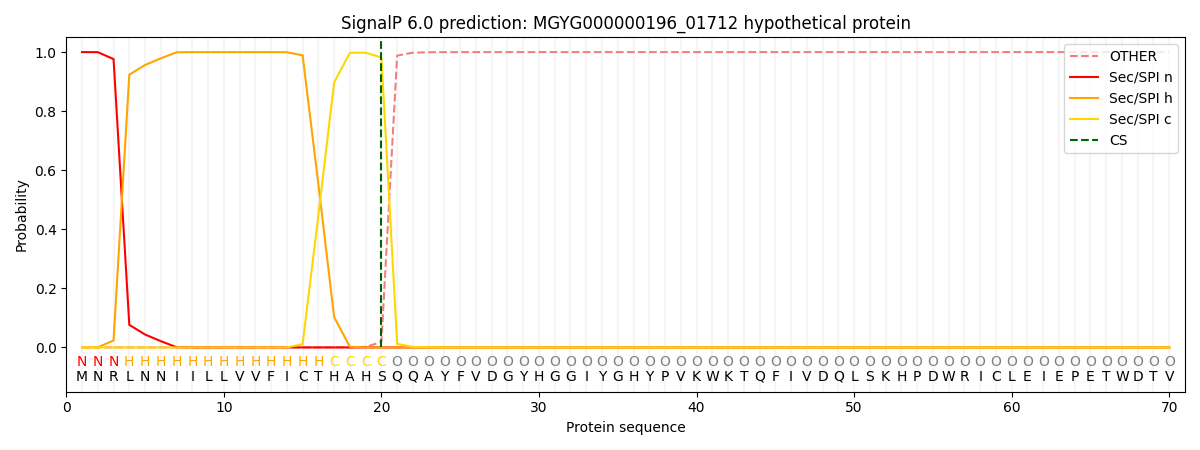
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000205 | 0.999232 | 0.000147 | 0.000140 | 0.000132 | 0.000127 |
