You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000373_00259
You are here: Home > Sequence: MGYG000000373_00259
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
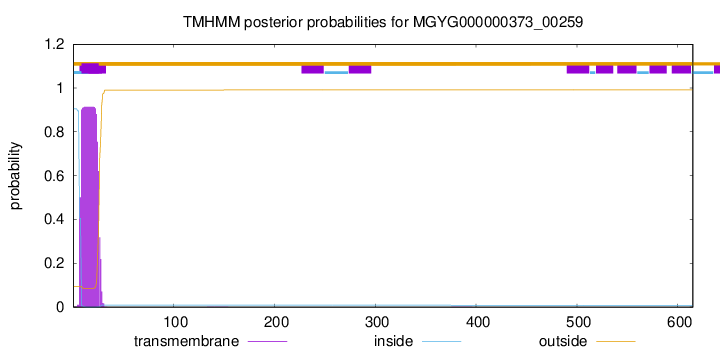
TMHMM annotations
Basic Information help
| Species | UMGS1537 sp900761045 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; UBA1212; UBA1255; UMGS1537; UMGS1537 sp900761045 | |||||||||||
| CAZyme ID | MGYG000000373_00259 | |||||||||||
| CAZy Family | GH163 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 24460; End: 26307 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH163 | 244 | 506 | 2.6e-90 | 0.9960159362549801 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam16126 | DUF4838 | 1.87e-109 | 237 | 506 | 1 | 263 | Domain of unknown function (DUF4838). This family consists of several uncharacterized proteins found in various Bacteroides and Chloroflexus species. The function of this family is unknown. |
| pfam03648 | Glyco_hydro_67N | 3.25e-08 | 58 | 151 | 24 | 119 | Glycosyl hydrolase family 67 N-terminus. Alpha-glucuronidases, components of an ensemble of enzymes central to the recycling of photosynthetic biomass, remove the alpha-1,2 linked 4-O-methyl glucuronic acid from xylans. This family represents the N-terminal region of alpha-glucuronidase. The N-terminal domain forms a two-layer sandwich, each layer being formed by a beta sheet of five strands. A further two helices form part of the interface with the central, catalytic, module (pfam07488). |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QOY90686.1 | 2.66e-117 | 43 | 571 | 15 | 528 |
| ASV73444.1 | 4.53e-115 | 43 | 566 | 24 | 537 |
| AIF26900.1 | 5.66e-108 | 47 | 507 | 6 | 477 |
| QQL44163.1 | 2.08e-106 | 44 | 566 | 9 | 523 |
| AIE83908.1 | 2.93e-102 | 56 | 587 | 22 | 528 |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as LIPO

| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000000 | 0.000005 | 1.000034 | 0.000000 | 0.000000 | 0.000000 |