You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000608_01761
You are here: Home > Sequence: MGYG000000608_01761
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Verrucomicrobiota; Lentisphaeria; Victivallales; Victivallaceae; UMGS1518; | |||||||||||
| CAZyme ID | MGYG000000608_01761 | |||||||||||
| CAZy Family | GH31 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 140; End: 2773 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH31 | 295 | 535 | 4.8e-43 | 0.5573770491803278 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| COG1501 | YicI | 5.25e-57 | 176 | 804 | 227 | 748 | Alpha-glucosidase, glycosyl hydrolase family GH31 [Carbohydrate transport and metabolism]. |
| cd06595 | GH31_u1 | 4.74e-48 | 206 | 478 | 1 | 294 | glycosyl hydrolase family 31 (GH31); uncharacterized subgroup. This family represents an uncharacterized GH31 enzyme subgroup found in bacteria and eukaryotes. Enzymes of the GH31 family possess a wide range of different hydrolytic activities including alpha-glucosidase (glucoamylase and sucrase-isomaltase), alpha-xylosidase, 6-alpha-glucosyltransferase, 3-alpha-isomaltosyltransferase and alpha-1,4-glucan lyase. All GH31 enzymes cleave a terminal carbohydrate moiety from a substrate that varies considerably in size, depending on the enzyme, and may be either a starch or a glycoprotein. |
| pfam01055 | Glyco_hydro_31 | 1.42e-46 | 186 | 726 | 1 | 442 | Glycosyl hydrolases family 31. Glycosyl hydrolases are key enzymes of carbohydrate metabolism. Family 31 comprises of enzymes that are, or similar to, alpha- galactosidases. |
| cd06589 | GH31 | 1.32e-35 | 207 | 477 | 1 | 264 | glycosyl hydrolase family 31 (GH31). GH31 enzymes occur in prokaryotes, eukaryotes, and archaea with a wide range of hydrolytic activities, including alpha-glucosidase (glucoamylase and sucrase-isomaltase), alpha-xylosidase, 6-alpha-glucosyltransferase, 3-alpha-isomaltosyltransferase and alpha-1,4-glucan lyase. All GH31 enzymes cleave a terminal carbohydrate moiety from a substrate that varies considerably in size, depending on the enzyme, and may be either a starch or a glycoprotein. In most cases, the pyranose moiety recognized in subsite -1 of the substrate binding site is an alpha-D-glucose, though some GH31 family members show a preference for alpha-D-xylose. Several GH31 enzymes can accommodate both glucose and xylose and different levels of discrimination between the two have been observed. Most characterized GH31 enzymes are alpha-glucosidases. In mammals, GH31 members with alpha-glucosidase activity are implicated in at least three distinct biological processes. The lysosomal acid alpha-glucosidase (GAA) is essential for glycogen degradation and a deficiency or malfunction of this enzyme causes glycogen storage disease II, also known as Pompe disease. In the endoplasmic reticulum, alpha-glucosidase II catalyzes the second step in the N-linked oligosaccharide processing pathway that constitutes part of the quality control system for glycoprotein folding and maturation. The intestinal enzymes sucrase-isomaltase (SI) and maltase-glucoamylase (MGAM) play key roles in the final stage of carbohydrate digestion, making alpha-glucosidase inhibitors useful in the treatment of type 2 diabetes. GH31 alpha-glycosidases are retaining enzymes that cleave their substrates via an acid/base-catalyzed, double-displacement mechanism involving a covalent glycosyl-enzyme intermediate. Two aspartic acid residues have been identified as the catalytic nucleophile and the acid/base, respectively. |
| pfam07691 | PA14 | 4.70e-27 | 553 | 672 | 4 | 126 | PA14 domain. This domain forms an insert in bacterial beta-glucosidases and is found in other glycosidases, glycosyltransferases, proteases, amidases, yeast adhesins, and bacterial toxins, including anthrax protective antigen (PA). The domain also occurs in a Dictyostelium prespore-cell-inducing factor Psi and in fibrocystin, the mammalian protein whose mutation leads to polycystic kidney and hepatic disease. The crystal structure of PA shows that this domain (named PA14 after its location in the PA20 pro-peptide) has a beta-barrel structure. The PA14 domain sequence suggests a binding function, rather than a catalytic role. The PA14 domain distribution is compatible with carbohydrate binding. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| AIE84875.1 | 2.36e-205 | 31 | 847 | 6 | 807 |
| ARN57252.1 | 1.23e-132 | 135 | 871 | 263 | 853 |
| AQQ09813.1 | 9.93e-125 | 135 | 833 | 263 | 817 |
| VEI51975.1 | 2.33e-49 | 30 | 592 | 9 | 612 |
| QCY31939.1 | 2.33e-49 | 30 | 592 | 9 | 612 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 7WJ9_A | 1.47e-49 | 41 | 533 | 34 | 555 | ChainA, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJ9_B Chain B, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJ9_C Chain C, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJ9_D Chain D, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJ9_E Chain E, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJ9_F Chain F, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJA_A Chain A, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJB_A Chain A, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WLG_A Chain A, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WLG_B Chain B, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WLG_C Chain C, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WLG_D Chain D, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WLG_E Chain E, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WLG_F Chain F, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363] |
| 7WJC_A | 1.58e-48 | 41 | 533 | 34 | 555 | ChainA, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJD_A Chain A, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJE_A Chain A, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363],7WJF_A Chain A, Alpha-xylosidase [Lactococcus lactis subsp. cremoris MG1363] |
| 6JR6_A | 1.02e-34 | 185 | 534 | 237 | 637 | Flavobacteriumjohnsoniae GH31 dextranase, FjDex31A [Flavobacterium johnsoniae UW101],6JR6_B Flavobacterium johnsoniae GH31 dextranase, FjDex31A [Flavobacterium johnsoniae UW101],6JR6_C Flavobacterium johnsoniae GH31 dextranase, FjDex31A [Flavobacterium johnsoniae UW101],6JR6_D Flavobacterium johnsoniae GH31 dextranase, FjDex31A [Flavobacterium johnsoniae UW101],6JR7_A Flavobacterium johnsoniae GH31 dextranase, FjDex31A, complexed with glucose [Flavobacterium johnsoniae UW101],6JR7_B Flavobacterium johnsoniae GH31 dextranase, FjDex31A, complexed with glucose [Flavobacterium johnsoniae UW101],6JR7_C Flavobacterium johnsoniae GH31 dextranase, FjDex31A, complexed with glucose [Flavobacterium johnsoniae UW101],6JR7_D Flavobacterium johnsoniae GH31 dextranase, FjDex31A, complexed with glucose [Flavobacterium johnsoniae UW101] |
| 6JR8_A | 7.25e-34 | 185 | 534 | 237 | 637 | Flavobacteriumjohnsoniae GH31 dextranase, FjDex31A, mutant D412A complexed with isomaltotriose [Flavobacterium johnsoniae UW101],6JR8_B Flavobacterium johnsoniae GH31 dextranase, FjDex31A, mutant D412A complexed with isomaltotriose [Flavobacterium johnsoniae UW101],6JR8_C Flavobacterium johnsoniae GH31 dextranase, FjDex31A, mutant D412A complexed with isomaltotriose [Flavobacterium johnsoniae UW101],6JR8_D Flavobacterium johnsoniae GH31 dextranase, FjDex31A, mutant D412A complexed with isomaltotriose [Flavobacterium johnsoniae UW101] |
| 5X7O_A | 1.14e-26 | 172 | 821 | 217 | 772 | Crystalstructure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase [Paenibacillus sp. 598K],5X7O_B Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase [Paenibacillus sp. 598K],5X7P_A Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase complexed with acarbose [Paenibacillus sp. 598K],5X7P_B Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase complexed with acarbose [Paenibacillus sp. 598K],5X7Q_A Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase complexed with maltohexaose [Paenibacillus sp. 598K],5X7Q_B Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase complexed with maltohexaose [Paenibacillus sp. 598K],5X7R_A Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase complexed with isomaltohexaose [Paenibacillus sp. 598K],5X7R_B Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase complexed with isomaltohexaose [Paenibacillus sp. 598K],5X7S_A Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase, terbium derivative [Paenibacillus sp. 598K],5X7S_B Crystal structure of Paenibacillus sp. 598K alpha-1,6-glucosyltransferase, terbium derivative [Paenibacillus sp. 598K] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| Q5AW25 | 4.58e-29 | 186 | 536 | 257 | 641 | Alpha-xylosidase OS=Emericella nidulans (strain FGSC A4 / ATCC 38163 / CBS 112.46 / NRRL 194 / M139) OX=227321 GN=agdD PE=1 SV=1 |
| B3PEE6 | 1.11e-24 | 186 | 533 | 234 | 620 | Oligosaccharide 4-alpha-D-glucosyltransferase OS=Cellvibrio japonicus (strain Ueda107) OX=498211 GN=agd31B PE=1 SV=1 |
| P96793 | 1.74e-24 | 186 | 536 | 237 | 624 | Alpha-xylosidase XylQ OS=Lactiplantibacillus pentosus OX=1589 GN=xylQ PE=1 SV=1 |
| Q9P999 | 6.50e-24 | 176 | 809 | 183 | 702 | Alpha-xylosidase OS=Saccharolobus solfataricus (strain ATCC 35092 / DSM 1617 / JCM 11322 / P2) OX=273057 GN=xylS PE=1 SV=1 |
| P31434 | 3.76e-23 | 196 | 585 | 233 | 686 | Alpha-xylosidase OS=Escherichia coli (strain K12) OX=83333 GN=yicI PE=1 SV=2 |
SignalP and Lipop Annotations help
This protein is predicted as SP
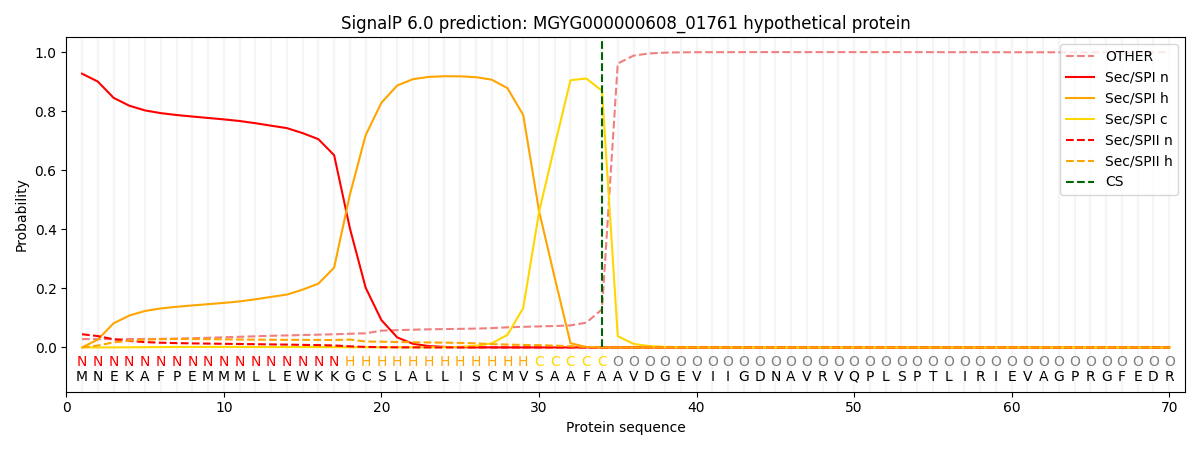
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.030795 | 0.919600 | 0.048442 | 0.000533 | 0.000299 | 0.000313 |
