You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000000788_02297
You are here: Home > Sequence: MGYG000000788_02297
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Bacteroides pyogenes | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Bacteroides; Bacteroides pyogenes | |||||||||||
| CAZyme ID | MGYG000000788_02297 | |||||||||||
| CAZy Family | GT80 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 88; End: 1554 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GT80 | 317 | 467 | 3.6e-32 | 0.41688654353562005 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam11477 | PM0188 | 1.88e-04 | 397 | 468 | 286 | 357 | Sialyltransferase PMO188. PMO188 is a sialyltransferase from P.multocida. It transfers sialic acid from cytidine 5'-monophosphonuraminic acid to an acceptor sugar. It has important catalytic residues such as Asp141, His311, Glu338, Ser355 and Ser356. |
| cd14948 | BACON | 0.001 | 38 | 117 | 6 | 83 | Bacteroidetes-Associated Carbohydrate-binding (putative) Often N-terminal (BACON) domain. The BACON domain is found in diverse domain architectures and accociated with a wide variety of domains, including carbohydrate-active enzymes and proteases. It was named for its suggested function of carbohydrate binding; the latter was inferred from domain architectures, sequence conservation, and phyletic distribution. However, recent experimental data suggest that its primary function in Bacteroides ovatus endo-xyloglucanase BoGH5A is to distance the catalytic module from the cell surface and confer additional mobility to the catalytic domain for attack of the polysaccharide. No evidence for a direct role in carbohydrate binding could be found in that case. The large majority of BACON domains are found in Bacteroidetes. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| BBL01105.1 | 3.26e-157 | 4 | 488 | 2 | 497 |
| BBL09010.1 | 5.22e-60 | 305 | 488 | 1 | 188 |
| BBL11802.1 | 5.22e-60 | 305 | 488 | 1 | 188 |
| BAF92026.1 | 2.48e-06 | 318 | 471 | 310 | 471 |
| BAF91416.1 | 9.81e-06 | 164 | 469 | 157 | 466 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 2Z4T_A | 9.07e-08 | 318 | 471 | 310 | 471 | CrystalStructure of Vibrionaceae Photobacterium sp. JT-ISH-224 2,6-sialyltransferase in a Ternary Complex with Donor Product CMP and Accepter Substrate Lactose [Photobacterium sp. JT-ISH-224] |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as LIPO
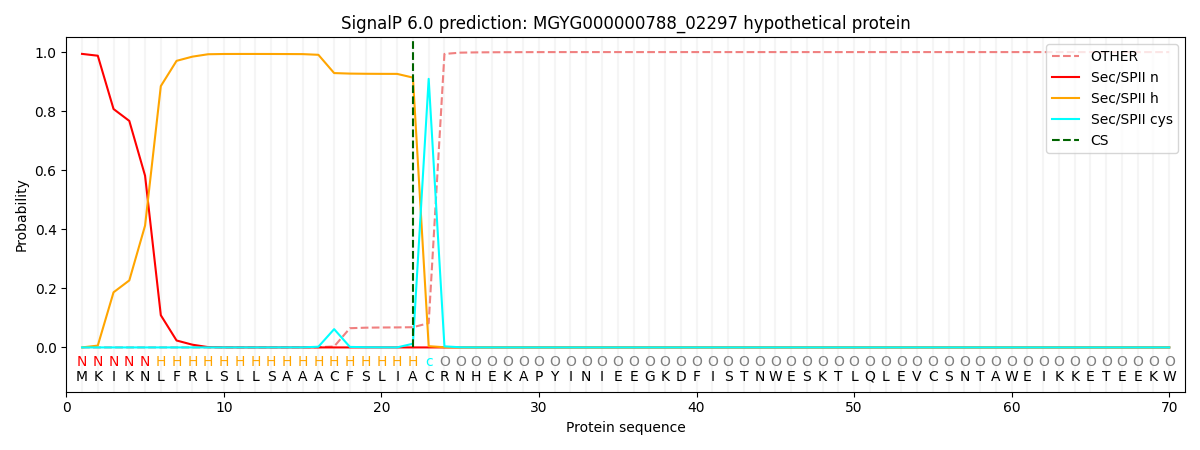
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000014 | 0.005835 | 0.994194 | 0.000002 | 0.000004 | 0.000003 |
