You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001206_00460
You are here: Home > Sequence: MGYG000001206_00460
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | UMGS1491 sp900554775 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; Oscillospirales; Acutalibacteraceae; UMGS1491; UMGS1491 sp900554775 | |||||||||||
| CAZyme ID | MGYG000001206_00460 | |||||||||||
| CAZy Family | GH24 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 78109; End: 79479 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH24 | 215 | 320 | 1e-18 | 0.7445255474452555 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd00737 | lyz_endolysin_autolysin | 7.40e-21 | 218 | 317 | 1 | 95 | endolysin and autolysin. The dsDNA phages of eubacteria use endolysins or muralytic enzymes in conjunction with hollin, a small membrane protein, to degrade the peptidoglycan found in bacterial cell walls. Similarly, bacteria produce autolysins to facilitate the biosynthesis of its cell wall heteropolymer peptidoglycan and cell division. Endolysins and autolysins are found in viruses and bacteria, respectively. Both endolysin and autolysin enzymes cleave the glycosidic beta 1,4-bonds between the N-acetylmuramic acid and the N-acetylglucosamine of the peptidoglycan. |
| pfam08239 | SH3_3 | 1.00e-14 | 336 | 388 | 1 | 54 | Bacterial SH3 domain. |
| COG3772 | RrrD | 1.80e-10 | 214 | 341 | 7 | 123 | Phage-related lysozyme (muramidase), GH24 family [Cell wall/membrane/envelope biogenesis]. |
| smart00287 | SH3b | 2.72e-08 | 327 | 381 | 1 | 55 | Bacterial SH3 domain homologues. |
| cd16901 | lyz_P1 | 3.21e-08 | 214 | 317 | 2 | 99 | P1 lysozyme Lyz-like proteins. Enterobacteria phage P1 lysozyme Lyz is secreted to the Escherichia coli periplasm where it is membrane bound and inactive. Activation involves the release from the membrane, an intramolecular thiol-disulfide isomerization and extensive structural rearrangement of the N-terminal region. The dsDNA phages of eubacteria use endolysins or muralytic enzymes in conjunction with hollin, a small membrane protein, to degrade the peptidoglycan found in bacterial cell walls. Similarly, bacteria produce autolysins to facilitate the biosynthesis of its cell wall heteropolymer peptidoglycan and cell division. Endolysins and autolysins are found in viruses and bacteria, respectively. Both endolysin and autolysin enzymes cleave the glycosidic beta 1,4-bonds between the N-acetylmuramic acid and the N-acetylglucosamine of the peptidoglycan. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QJA51100.1 | 1.72e-12 | 214 | 320 | 3 | 104 |
| QJI00463.1 | 5.96e-12 | 214 | 320 | 3 | 104 |
| AWB43148.1 | 2.44e-10 | 214 | 322 | 495 | 602 |
| QVG63981.1 | 1.49e-09 | 212 | 324 | 75 | 184 |
| QQN37305.1 | 2.43e-09 | 213 | 340 | 2 | 117 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 6ET6_A | 5.60e-07 | 212 | 316 | 50 | 152 | ChainA, Lysozyme [Acinetobacter baumannii] |
| 2KRS_A | 5.30e-06 | 329 | 393 | 2 | 65 | SolutionNMR structure of SH3 domain from CPF_0587 (fragment 415-479) from Clostridium perfringens. Northeast Structural Genomics Consortium (NESG) Target CpR74A. [Clostridium perfringens] |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as SP
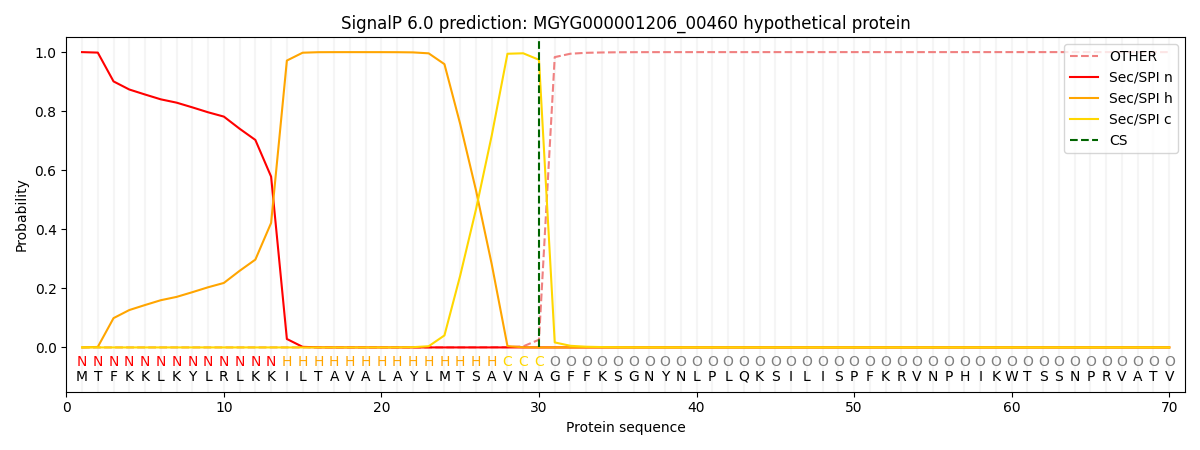
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000408 | 0.998884 | 0.000198 | 0.000175 | 0.000159 | 0.000143 |
