You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001340_00698
You are here: Home > Sequence: MGYG000001340_00698
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Weissella paramesenteroides | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes; Bacilli; Lactobacillales; Lactobacillaceae; Weissella; Weissella paramesenteroides | |||||||||||
| CAZyme ID | MGYG000001340_00698 | |||||||||||
| CAZy Family | CBM50 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 94806; End: 96230 Strand: - | |||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam00877 | NLPC_P60 | 6.41e-25 | 363 | 458 | 3 | 91 | NlpC/P60 family. The function of this domain is unknown. It is found in several lipoproteins. |
| COG0791 | Spr | 5.80e-20 | 340 | 459 | 64 | 183 | Cell wall-associated hydrolase, NlpC family [Cell wall/membrane/envelope biogenesis]. |
| PRK13914 | PRK13914 | 4.81e-17 | 31 | 450 | 51 | 460 | invasion associated endopeptidase. |
| NF033742 | NlpC_p60_RipB | 1.04e-11 | 358 | 456 | 85 | 189 | NlpC/P60 family peptidoglycan endopeptidase RipB. |
| cd00118 | LysM | 3.15e-11 | 164 | 207 | 2 | 45 | Lysin Motif is a small domain involved in binding peptidoglycan. LysM, a small globular domain with approximately 40 amino acids, is a widespread protein module involved in binding peptidoglycan in bacteria and chitin in eukaryotes. The domain was originally identified in enzymes that degrade bacterial cell walls, but proteins involved in many other biological functions also contain this domain. It has been reported that the LysM domain functions as a signal for specific plant-bacteria recognition in bacterial pathogenesis. Many of these enzymes are modular and are composed of catalytic units linked to one or several repeats of LysM domains. LysM domains are found in bacteria and eukaryotes. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QPI45904.1 | 7.15e-300 | 1 | 474 | 1 | 474 |
| ATF40903.1 | 7.15e-300 | 1 | 474 | 1 | 474 |
| APS42221.1 | 6.78e-199 | 1 | 474 | 1 | 482 |
| QDJ59119.1 | 2.70e-124 | 1 | 473 | 1 | 470 |
| QEA56411.1 | 4.33e-123 | 1 | 473 | 1 | 470 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 6B8C_A | 1.59e-16 | 348 | 458 | 27 | 131 | Crystalstructure of NlpC/p60 domain of peptidoglycan hydrolase SagA [Enterococcus faecium] |
| 3PBC_A | 1.51e-08 | 362 | 465 | 98 | 206 | ChainA, Invasion Protein [Mycobacterium tuberculosis] |
| 3NE0_A | 1.51e-08 | 362 | 465 | 98 | 206 | Structureand functional regulation of RipA, a mycobacterial enzyme essential for daughter cell separation [Mycobacterium tuberculosis H37Rv] |
| 2XIV_A | 2.58e-08 | 362 | 465 | 93 | 201 | Structureof Rv1477, hypothetical invasion protein of Mycobacterium tuberculosis [Mycobacterium tuberculosis H37Rv] |
| 4Q4T_A | 3.49e-08 | 362 | 465 | 356 | 464 | Structureof the Resuscitation Promoting Factor Interacting protein RipA mutated at E444 [Mycobacterium tuberculosis H37Rv] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| P13692 | 4.94e-14 | 348 | 458 | 401 | 505 | Protein P54 OS=Enterococcus faecium OX=1352 PE=3 SV=2 |
| P39043 | 3.34e-09 | 347 | 458 | 151 | 249 | Dipeptidyl-peptidase 6 OS=Lysinibacillus sphaericus OX=1421 PE=1 SV=1 |
| P96645 | 1.66e-08 | 350 | 452 | 212 | 309 | Probable endopeptidase YddH OS=Bacillus subtilis (strain 168) OX=224308 GN=yddH PE=3 SV=1 |
| O35010 | 1.83e-07 | 362 | 461 | 186 | 281 | Gamma-D-glutamyl-L-lysine dipeptidyl-peptidase OS=Bacillus subtilis (strain 168) OX=224308 GN=ykfC PE=1 SV=2 |
| O53168 | 3.35e-07 | 362 | 465 | 356 | 464 | Peptidoglycan endopeptidase RipA OS=Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv) OX=83332 GN=ripA PE=1 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
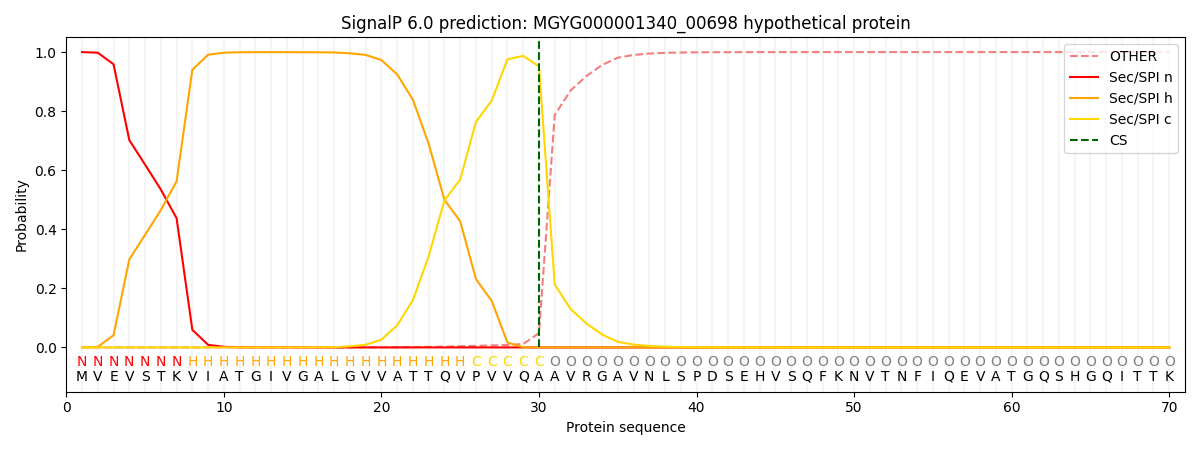
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000756 | 0.998408 | 0.000200 | 0.000232 | 0.000200 | 0.000190 |
