You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001404_04330
You are here: Home > Sequence: MGYG000001404_04330
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Paenibacillus_L senegalensis | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes; Bacilli; Paenibacillales; Paenibacillaceae; Paenibacillus_L; Paenibacillus_L senegalensis | |||||||||||
| CAZyme ID | MGYG000001404_04330 | |||||||||||
| CAZy Family | CBM50 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 341559; End: 343424 Strand: - | |||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| PRK09419 | PRK09419 | 3.42e-125 | 41 | 515 | 660 | 1149 | multifunctional 2',3'-cyclic-nucleotide 2'-phosphodiesterase/3'-nucleotidase/5'-nucleotidase. |
| COG0737 | UshA | 6.76e-121 | 16 | 515 | 1 | 517 | 2',3'-cyclic-nucleotide 2'-phosphodiesterase/5'- or 3'-nucleotidase, 5'-nucleotidase family [Nucleotide transport and metabolism, Defense mechanisms]. |
| PRK09558 | ushA | 4.36e-120 | 1 | 503 | 1 | 539 | bifunctional UDP-sugar hydrolase/5'-nucleotidase periplasmic precursor; Reviewed |
| cd07408 | MPP_SA0022_N | 7.47e-81 | 42 | 283 | 1 | 246 | Staphylococcus aureus SA0022 and related proteins, N-terminal metallophosphatase domain. SA0022 is an uncharacterized Staphylococcus aureus UshA-like protein with two putative domains, an N-terminal metallophosphatase domain and a C-terminal nucleotidase domain. SA0022 also contains a putative C-terminal cell wall anchor domain. The N-terminal metallophosphatase domain belongs to a large superfamily of distantly related metallophosphatases (MPPs) that includes: Mre11/SbcD-like exonucleases, Dbr1-like RNA lariat debranching enzymes, YfcE-like phosphodiesterases, purple acid phosphatases (PAPs), YbbF-like UDP-2,3-diacylglucosamine hydrolases, and acid sphingomyelinases (ASMases). MPPs are functionally diverse, but all share a conserved domain with an active site consisting of two metal ions (usually manganese, iron, or zinc) coordinated with octahedral geometry by a cage of histidine, aspartate, and asparagine residues. The conserved domain is a double beta-sheet sandwich with a di-metal active site made up of residues located at the C-terminal side of the sheets. This domain is thought to allow for productive metal coordination. |
| cd00845 | MPP_UshA_N_like | 1.38e-75 | 42 | 282 | 1 | 242 | Escherichia coli UshA-like family, N-terminal metallophosphatase domain. This family includes the bacterial enzyme UshA, and related enzymes including SoxB, CpdB, YhcR, and CD73. All members have a similar domain architecture which includes an N-terminal metallophosphatase domain and a C-terminal nucleotidase domain. The N-terminal metallophosphatase domain belongs to a large superfamily of distantly related metallophosphatases (MPPs) that includes: Mre11/SbcD-like exonucleases, Dbr1-like RNA lariat debranching enzymes, YfcE-like phosphodiesterases, purple acid phosphatases (PAPs), YbbF-like UDP-2,3-diacylglucosamine hydrolases, and acid sphingomyelinases (ASMases). MPPs are functionally diverse, but all share a conserved domain with an active site consisting of two metal ions (usually manganese, iron, or zinc) coordinated with octahedral geometry by a cage of histidine, aspartate, and asparagine residues. The conserved domain is a double beta-sheet sandwich with a di-metal active site made up of residues located at the C-terminal side of the sheets. This domain is thought to allow for productive metal coordination. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| AGA59602.1 | 7.37e-244 | 12 | 621 | 10 | 601 |
| AEI45885.1 | 4.78e-215 | 24 | 621 | 31 | 592 |
| AFH65854.2 | 8.33e-215 | 24 | 621 | 31 | 598 |
| AFC33532.1 | 2.73e-214 | 24 | 621 | 31 | 592 |
| AFK65438.1 | 2.29e-212 | 24 | 621 | 22 | 579 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 2Z1A_A | 6.81e-75 | 27 | 515 | 15 | 534 | Crystalstructure of 5'-nucleotidase precursor from Thermus thermophilus HB8 [Thermus thermophilus HB8] |
| 1HP1_A | 3.96e-62 | 42 | 503 | 9 | 504 | 5'-Nucleotidase(Open Form) Complex With Atp [Escherichia coli] |
| 1USH_A | 1.14e-60 | 28 | 503 | 18 | 538 | 5'-NucleotidaseFrom E. Coli [Escherichia coli K-12],2USH_A 5'-Nucleotidase From E. Coli [Escherichia coli K-12],2USH_B 5'-Nucleotidase From E. Coli [Escherichia coli K-12] |
| 1HO5_A | 3.47e-60 | 42 | 503 | 9 | 513 | 5'-Nucleotidase(E. Coli) In Complex With Adenosine And Phosphate [Escherichia coli],1HO5_B 5'-Nucleotidase (E. Coli) In Complex With Adenosine And Phosphate [Escherichia coli],1HPU_A 5'-Nucleotidase (Closed Form), Complex With Ampcp [Escherichia coli],1HPU_B 5'-Nucleotidase (Closed Form), Complex With Ampcp [Escherichia coli],1HPU_C 5'-Nucleotidase (Closed Form), Complex With Ampcp [Escherichia coli],1HPU_D 5'-Nucleotidase (Closed Form), Complex With Ampcp [Escherichia coli] |
| 1OI8_A | 1.08e-59 | 42 | 503 | 9 | 513 | 5'-Nucleotidase(E. coli) with an Engineered Disulfide Bridge (P90C, L424C) [Escherichia coli],1OI8_B 5'-Nucleotidase (E. coli) with an Engineered Disulfide Bridge (P90C, L424C) [Escherichia coli] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| A9BJC1 | 7.13e-88 | 42 | 509 | 24 | 496 | Mannosylglucosyl-3-phosphoglycerate phosphatase OS=Petrotoga mobilis (strain DSM 10674 / SJ95) OX=403833 GN=mggB PE=1 SV=1 |
| O34313 | 2.10e-73 | 42 | 509 | 669 | 1174 | Trifunctional nucleotide phosphoesterase protein YfkN OS=Bacillus subtilis (strain 168) OX=224308 GN=yfkN PE=1 SV=1 |
| Q9KQ30 | 9.25e-61 | 11 | 503 | 9 | 541 | 5'-nucleotidase OS=Vibrio cholerae serotype O1 (strain ATCC 39315 / El Tor Inaba N16961) OX=243277 GN=nutA PE=3 SV=1 |
| P07024 | 6.22e-60 | 28 | 503 | 18 | 538 | Protein UshA OS=Escherichia coli (strain K12) OX=83333 GN=ushA PE=1 SV=2 |
| P29240 | 7.70e-60 | 42 | 509 | 32 | 546 | 5'-nucleotidase OS=Diplobatis ommata OX=1870830 PE=2 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
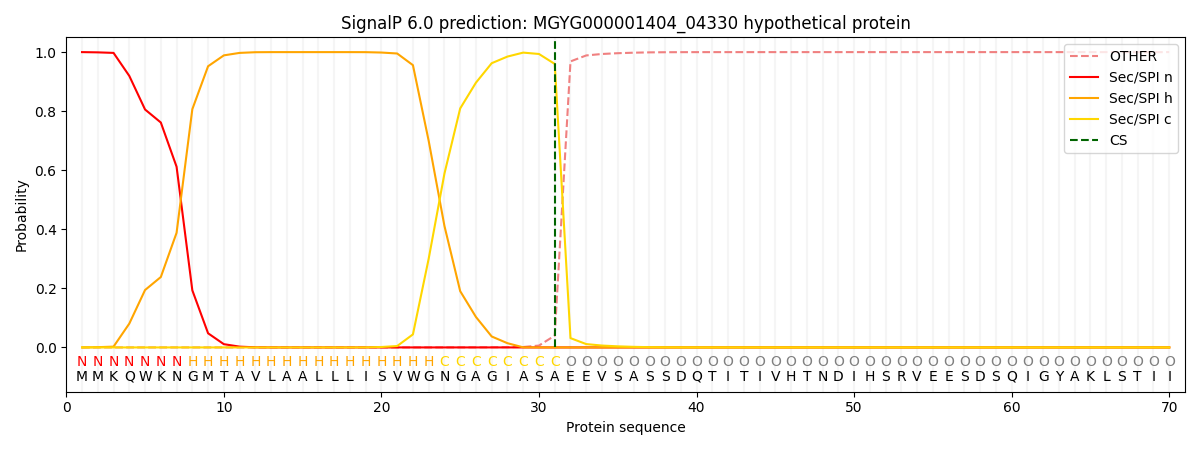
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000557 | 0.998557 | 0.000224 | 0.000255 | 0.000209 | 0.000186 |
