You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000001633_01237
You are here: Home > Sequence: MGYG000001633_01237
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | RC9 sp000435075 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; UBA932; RC9; RC9 sp000435075 | |||||||||||
| CAZyme ID | MGYG000001633_01237 | |||||||||||
| CAZy Family | GH16 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 22503; End: 25187 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH16 | 539 | 835 | 1.2e-19 | 0.9894179894179894 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam07980 | SusD_RagB | 4.41e-40 | 295 | 537 | 2 | 273 | SusD family. This domain is found in bacterial cell surface proteins such SusD and SusD-like proteins, as as well RagB, outer membrane surface receptor antigen. Bacteroidetes, one of the two dominant bacterial phyla in the human gut, are Gram-negative saccharolytic microorganisms that utilize a diverse array of glycans. Hence, they express starch-utilization system (Sus) for glycan uptake. SusD has 551 amino acids, and is almost entirely alpha-helical, with 22 alpha-helices, eight of which form 4 tetra-trico peptide repeats (TPRs: helix-turn-helix motifs involved in protein-protein interactions). The four TPRs pack together to create a right-handed super-helix. This is predicted to mediate the formation of SusD and SusC porin complex at the cell surface. The interaction between SusC and TPR1/TPR2 region of SusD is predicted to be of functional importance since it allows SusD to be in position for oligosaccharide capture from other Sus lipoproteins and delivery of these glycans to the SusC porin. The non-TPR containing portion of SusD is where starch binding occurs. The binding site is a shallow surface cavity located on top of TPR1. SusD homologs such as SusD-like proteins have a critical role in carbohydrate acquisition. Both SusD and its homologs, contain about 15-20 residues at the N-terminus that might be a flexible linker region, anchoring the protein to the membrane and the glycan-binding domain. Other homologs to SusD have been examined in Porphyromonas gingivalis such as RagB, an immunodominant outer-membrane surface receptor antigen. Structural characterization of RagB shows substantial similarity with Bacteroides thetaiotaomicron SusD (i.e alpha-helices and TPR regions). Based on this structural similarity, functional studies suggest that, RagB binding of glycans occurs at pockets on the molecular surface that are distinct from those of SusD. |
| cd08977 | SusD | 1.07e-27 | 47 | 494 | 8 | 358 | starch binding outer membrane protein SusD. SusD-like proteins from Bacteroidetes, members of the human distal gut microbiota, are part of the starch utilization system (Sus). Sus is one of the large clusters of glycosyl hydrolases, called polysaccharide utilization loci (PULs), which play an important role in polysaccharide recognition and uptake, and it is needed for growth on amylose, amylopectin, pullulan, and maltooligosaccharides. SusD, together with SusC, a predicted beta-barrel porin, forms the minimum outer-membrane starch-binding complex. The adult human distal gut microbiota is essential for digestion of a large variety of dietary polysaccharides, for which humans lack the necessary glycosyl hydrolases. |
| cd08023 | GH16_laminarinase_like | 6.49e-15 | 541 | 835 | 3 | 235 | Laminarinase, member of the glycosyl hydrolase family 16. Laminarinase, also known as glucan endo-1,3-beta-D-glucosidase, is a glycosyl hydrolase family 16 member that hydrolyzes 1,3-beta-D-glucosidic linkages in 1,3-beta-D-glucans such as laminarins, curdlans, paramylons, and pachymans, with very limited action on mixed-link (1,3-1,4-)-beta-D-glucans. |
| cd00413 | Glyco_hydrolase_16 | 5.40e-09 | 541 | 835 | 1 | 210 | glycosyl hydrolase family 16. The O-Glycosyl hydrolases are a widespread group of enzymes that hydrolyse the glycosidic bond between two or more carbohydrates, or between a carbohydrate and a non-carbohydrate moiety. A glycosyl hydrolase classification system based on sequence similarity has led to the definition of more than 95 different families inlcuding glycosyl hydrolase family 16. Family 16 includes lichenase, xyloglucan endotransglycosylase (XET), beta-agarase, kappa-carrageenase, endo-beta-1,3-glucanase, endo-beta-1,3-1,4-glucanase, and endo-beta-galactosidase, all of which have a conserved jelly roll fold with a deep active site channel harboring the catalytic residues. |
| pfam14322 | SusD-like_3 | 6.23e-09 | 94 | 231 | 69 | 185 | Starch-binding associating with outer membrane. SusD is a secreted polysaccharide-binding protein with an N-terminal lipid moiety that allows it to associate with the outer membrane. SusD probably mediates xyloglucan-binding prior to xyloglucan transport in the periplasm for degradation. This domain is found N-terminal to pfam07980. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QRQ57561.1 | 0.0 | 7 | 893 | 9 | 887 |
| QDH56574.1 | 0.0 | 7 | 893 | 9 | 887 |
| ALJ45968.1 | 0.0 | 7 | 893 | 9 | 887 |
| SCV10319.1 | 1.15e-226 | 407 | 893 | 2 | 478 |
| QDH56573.1 | 2.96e-70 | 520 | 883 | 16 | 374 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 3QNK_A | 8.63e-42 | 93 | 500 | 71 | 466 | Crystalstructure of a SusD-like protein (BF3747) from Bacteroides fragilis NCTC 9343 at 2.70 A resolution [Bacteroides fragilis NCTC 9343],3QNK_B Crystal structure of a SusD-like protein (BF3747) from Bacteroides fragilis NCTC 9343 at 2.70 A resolution [Bacteroides fragilis NCTC 9343],3QNK_C Crystal structure of a SusD-like protein (BF3747) from Bacteroides fragilis NCTC 9343 at 2.70 A resolution [Bacteroides fragilis NCTC 9343],3QNK_D Crystal structure of a SusD-like protein (BF3747) from Bacteroides fragilis NCTC 9343 at 2.70 A resolution [Bacteroides fragilis NCTC 9343] |
| 7BLL_A | 6.53e-31 | 85 | 521 | 77 | 603 | ChainA, SusD homolog [Bacteroides thetaiotaomicron VPI-5482] |
| 5CJZ_A | 2.45e-29 | 96 | 506 | 85 | 573 | BT4246with galactose [Bacteroides thetaiotaomicron VPI-5482],5CK0_A Bt4246 [Bacteroides thetaiotaomicron VPI-5482] |
| 3I4G_A | 2.65e-29 | 101 | 507 | 92 | 474 | CRYSTALSTRUCTURE OF A SUSD-LIKE CARBOHYDRATE BINDING PROTEIN (BF0978) FROM BACTEROIDES FRAGILIS NCTC 9343 AT 1.35 A RESOLUTION [Bacteroides fragilis NCTC 9343] |
| 5CK1_A | 4.72e-29 | 96 | 506 | 100 | 588 | selenomethionineBT4246 [Bacteroides thetaiotaomicron VPI-5482] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| T2KNB8 | 5.75e-20 | 7 | 496 | 6 | 477 | SusD-like protein P25 OS=Formosa agariphila (strain DSM 15362 / KCTC 12365 / LMG 23005 / KMM 3901 / M-2Alg 35-1) OX=1347342 GN=BN863_22140 PE=2 SV=1 |
| T2KN63 | 3.45e-17 | 100 | 502 | 102 | 467 | SusD-like protein P2 OS=Formosa agariphila (strain DSM 15362 / KCTC 12365 / LMG 23005 / KMM 3901 / M-2Alg 35-1) OX=1347342 GN=BN863_21910 PE=2 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as LIPO
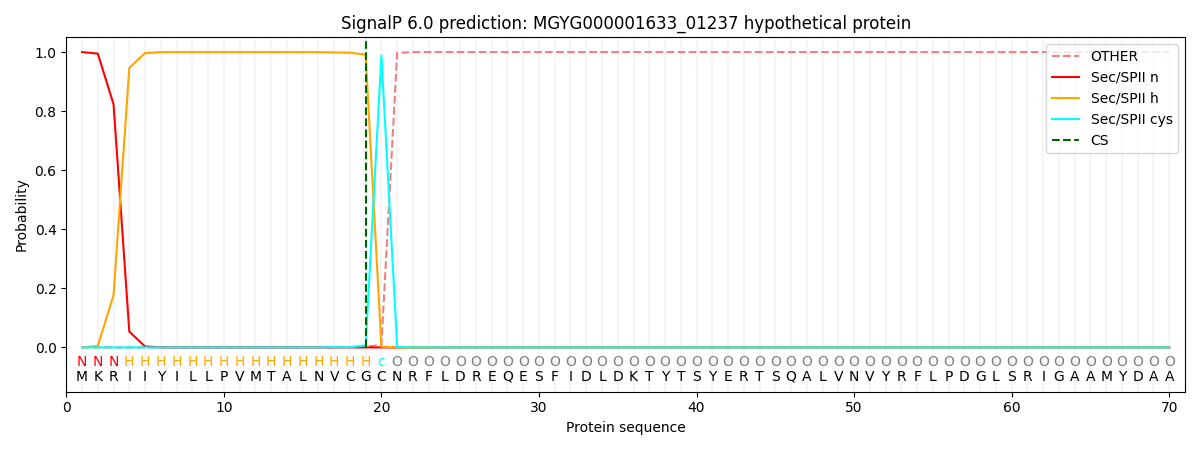
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000000 | 0.000006 | 1.000059 | 0.000000 | 0.000000 | 0.000000 |
