You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000002331_00451
You are here: Home > Sequence: MGYG000002331_00451
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Vibrio parahaemolyticus | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Proteobacteria; Gammaproteobacteria; Enterobacterales; Vibrionaceae; Vibrio; Vibrio parahaemolyticus | |||||||||||
| CAZyme ID | MGYG000002331_00451 | |||||||||||
| CAZy Family | GH0 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 457657; End: 460536 Strand: + | |||||||||||
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| pfam06202 | GDE_C | 2.98e-09 | 332 | 617 | 73 | 370 | Amylo-alpha-1,6-glucosidase. This family includes human glycogen branching enzyme AGL. This enzyme contains a number of distinct catalytic activities. It has been shown for the yeast homolog GDB1 that mutations in this region disrupt the enzymes Amylo-alpha-1,6-glucosidase (EC:3.2.1.33). |
| cd02861 | E_set_pullulanase_like | 1.09e-07 | 22 | 84 | 1 | 56 | Early set domain associated with the catalytic domain of pullulanase-like proteins. E or "early" set domains are associated with the catalytic domain of pullulanase at either the N-terminal or C-terminal end, and in a few instances at both ends. Pullulanase (also called dextrinase or alpha-dextrin endo-1,6-alpha glucosidase) is an enzyme with action similar to that of isoamylase; it cleaves 1,6-alpha-glucosidic linkages in pullulan, amylopectin, and glycogen, and in alpha-and beta-amylase limit-dextrins of amylopectin and glycogen. The E set domain of pullulanase may be related to the immunoglobulin and/or fibronectin type III superfamilies. These domains are associated with different types of catalytic domains at either the N-terminal or C-terminal end and may be involved in homodimeric/tetrameric/dodecameric interactions. Members of this family include members of the alpha amylase family, sialidase, galactose oxidase, cellulase, cellulose, hyaluronate lyase, chitobiase, and chitinase. This domain is also a member of the CBM48 (Carbohydrate Binding Module 48) family whose members include maltooligosyl trehalose synthase, starch branching enzyme, glycogen branching enzyme, glycogen debranching enzyme, isoamylase, and the beta subunit of AMP-activated protein kinase. |
| pfam17389 | Bac_rhamnosid6H | 2.36e-04 | 391 | 475 | 124 | 223 | Bacterial alpha-L-rhamnosidase 6 hairpin glycosidase domain. This family consists of bacterial rhamnosidase A and B enzymes. L-Rhamnose is abundant in biomass as a common constituent of glycolipids and glycosides, such as plant pigments, pectic polysaccharides, gums or biosurfactants. Some rhamnosides are important bioactive compounds. For example, terpenyl glycosides, the glycosidic precursor of aromatic terpenoids, act as important flavouring substances in grapes. Other rhamnosides act as cytotoxic rhamnosylated terpenoids, as signal substances in plants or play a role in the antigenicity of pathogenic bacteria. |
| COG4354 | COG4354 | 0.001 | 363 | 523 | 462 | 626 | Uncharacterized protein, contains GBA2_N and DUF608 domains [Function unknown]. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QBQ36601.1 | 6.27e-205 | 190 | 957 | 40 | 818 |
| AVR98316.1 | 7.53e-189 | 228 | 957 | 76 | 815 |
| QCP11581.1 | 1.68e-188 | 188 | 955 | 35 | 828 |
| AQT69211.1 | 2.89e-163 | 183 | 958 | 35 | 840 |
| AKT39344.1 | 1.13e-150 | 194 | 958 | 99 | 896 |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as LIPO
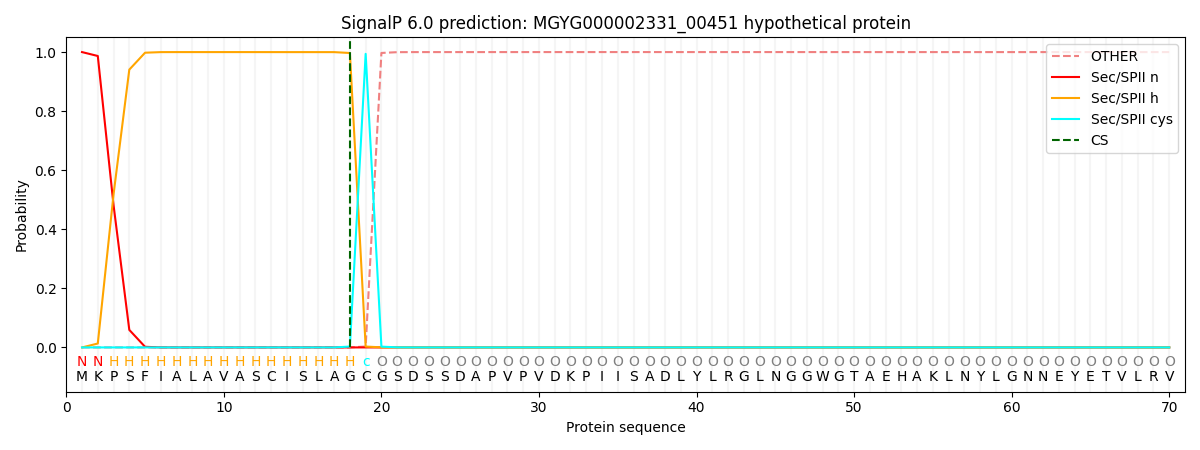
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000000 | 0.000000 | 1.000060 | 0.000000 | 0.000000 | 0.000000 |
