You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000002877_01099
You are here: Home > Sequence: MGYG000002877_01099
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Prevotella sp900550365 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Bacteroidota; Bacteroidia; Bacteroidales; Bacteroidaceae; Prevotella; Prevotella sp900550365 | |||||||||||
| CAZyme ID | MGYG000002877_01099 | |||||||||||
| CAZy Family | CE3 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 14093; End: 15457 Strand: + | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| CE3 | 32 | 221 | 2.4e-42 | 0.9896907216494846 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd01833 | XynB_like | 1.93e-26 | 31 | 221 | 1 | 157 | SGNH_hydrolase subfamily, similar to Ruminococcus flavefaciens XynB. Most likely a secreted hydrolase with xylanase activity. SGNH hydrolases are a diverse family of lipases and esterases. The tertiary fold of the enzyme is substantially different from that of the alpha/beta hydrolase family and unique among all known hydrolases; its active site closely resembles the Ser-His-Asp(Glu) triad found in other serine hydrolases. |
| pfam13472 | Lipase_GDSL_2 | 1.96e-17 | 36 | 209 | 2 | 173 | GDSL-like Lipase/Acylhydrolase family. This family of presumed lipases and related enzymes are similar to pfam00657. |
| cd00229 | SGNH_hydrolase | 1.35e-14 | 33 | 220 | 1 | 187 | SGNH_hydrolase, or GDSL_hydrolase, is a diverse family of lipases and esterases. The tertiary fold of the enzyme is substantially different from that of the alpha/beta hydrolase family and unique among all known hydrolases; its active site closely resembles the typical Ser-His-Asp(Glu) triad from other serine hydrolases, but may lack the carboxlic acid. |
| COG0657 | Aes | 1.94e-08 | 254 | 437 | 72 | 303 | Acetyl esterase/lipase [Lipid transport and metabolism]. |
| pfam00657 | Lipase_GDSL | 3.67e-08 | 33 | 214 | 1 | 221 | GDSL-like Lipase/Acylhydrolase. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| BAX78799.1 | 2.37e-117 | 25 | 447 | 32 | 490 |
| ATC65549.1 | 4.20e-113 | 33 | 430 | 40 | 474 |
| QOV88633.1 | 5.33e-65 | 10 | 227 | 9 | 228 |
| QNN20991.1 | 4.17e-52 | 33 | 221 | 32 | 222 |
| AFZ21551.1 | 4.41e-25 | 6 | 234 | 15 | 249 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 5AO9_A | 7.11e-19 | 236 | 448 | 18 | 275 | Thestructure of a novel thermophilic esterase from the Planctomycetes species, Thermogutta terrifontis, Est2-native [Thermogutta terrifontis],5AOA_A The structure of a novel thermophilic esterase from the Planctomycetes species, Thermogutta terrifontis, Est2-Propionate bound [Thermogutta terrifontis],5AOB_A The structure of a novel thermophilic esterase from the Planctomycetes species, Thermogutta terrifontis, Est2-butyrate bound [Thermogutta terrifontis],5AOC_A The structure of a novel thermophilic esterase from the Planctomycetes species, Thermogutta terrifontis, Est2-valerate bound [Thermogutta terrifontis] |
| 7BFN_A | 7.22e-19 | 236 | 448 | 19 | 276 | ChainA, Esterase [Thermogutta terrifontis] |
| 7BFO_A | 3.32e-18 | 236 | 448 | 19 | 276 | ChainA, Esterase [Thermogutta terrifontis],7BFR_A Chain A, Esterase [Thermogutta terrifontis],7BFT_A Chain A, Esterase [Thermogutta terrifontis],7BFU_A Chain A, Esterase [Thermogutta terrifontis],7BFV_A Chain A, Esterase [Thermogutta terrifontis] |
| 4YPV_A | 9.79e-09 | 255 | 369 | 109 | 229 | High-resolutionstructure of a metagenome-derived esterase Est8 [Parvibaculum] |
| 4Q3K_A | 1.43e-06 | 261 | 423 | 54 | 224 | Crystalstructure of MGS-M1, an alpha/beta hydrolase enzyme from a Medee basin deep-sea metagenome library [unidentified],4Q3K_B Crystal structure of MGS-M1, an alpha/beta hydrolase enzyme from a Medee basin deep-sea metagenome library [unidentified] |
Swiss-Prot Hits help
SignalP and Lipop Annotations help
This protein is predicted as SP
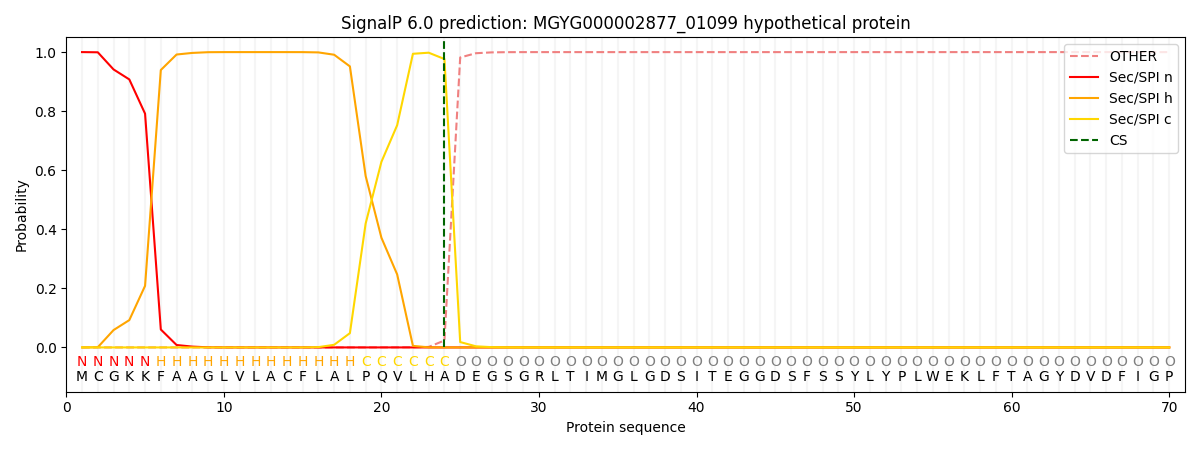
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000256 | 0.998943 | 0.000230 | 0.000187 | 0.000188 | 0.000170 |
