You are browsing environment: HUMAN GUT
CAZyme Information: MGYG000004667_02283
You are here: Home > Sequence: MGYG000004667_02283
Basic Information |
Genomic context |
Full Sequence |
Enzyme annotations |
CAZy signature domains |
CDD domains |
CAZyme hits |
PDB hits |
Swiss-Prot hits |
SignalP and Lipop annotations |
TMHMM annotations
Basic Information help
| Species | Clostridium beijerinckii | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lineage | Bacteria; Firmicutes_A; Clostridia; Clostridiales; Clostridiaceae; Clostridium; Clostridium beijerinckii | |||||||||||
| CAZyme ID | MGYG000004667_02283 | |||||||||||
| CAZy Family | GH16 | |||||||||||
| CAZyme Description | hypothetical protein | |||||||||||
| CAZyme Property |
|
|||||||||||
| Genome Property |
|
|||||||||||
| Gene Location | Start: 96219; End: 97811 Strand: - | |||||||||||
CAZyme Signature Domains help
| Family | Start | End | Evalue | family coverage |
|---|---|---|---|---|
| GH16 | 81 | 319 | 1.2e-58 | 0.9955357142857143 |
| CBM9 | 359 | 525 | 2.8e-29 | 0.9725274725274725 |
CDD Domains download full data without filtering help
| Cdd ID | Domain | E-Value | qStart | qEnd | sStart | sEnd | Domain Description |
|---|---|---|---|---|---|---|---|
| cd02178 | GH16_beta_agarase | 8.08e-60 | 64 | 320 | 9 | 258 | Beta-agarase, member of glycosyl hydrolase family 16. Beta-agarase is a glycosyl hydrolase family 16 (GH16) member that hydrolyzes the internal beta-1,4-linkage of agarose, a hydrophilic polysaccharide found in the cell wall of Rhodophyceaea, marine red algae. Agarose is a linear chain of galactose units linked by alternating L-alpha-1,3- and D-beta-1,4-linkages that are additionally modified by a 3,6-anhydro-bridge. Agarose forms thermo-reversible gels that are widely used in the food industry or as a laboratory medium. While beta-agarases are also found in two other families derived from the sequence-based classification of glycosyl hydrolases (GH50, and GH86) the GH16 members are most abundant. This domain adopts a curved beta-sandwich conformation, with a tunnel-shaped active site cavity, referred to as a jellyroll fold. |
| cd00005 | CBM9_like_1 | 2.46e-27 | 342 | 526 | 1 | 185 | DOMON-like type 9 carbohydrate binding module of xylanases. Family 9 carbohydrate-binding modules (CBM9) play a role in the microbial degradation of cellulose and hemicellulose (materials found in plants). The domain has previously been called cellulose-binding domain. The polysaccharide binding sites of CBMs with available 3D structure have been found to be either flat surfaces with interactions formed by predominantly aromatic residues (tryptophan and tyrosine), or extended shallow grooves. The CBM9 domain frequently occurs in tandem repeats; members found in this subfamily typically co-occur with glycosyl hydrolase family 10 domains and are annotated as endo-1,4-beta-xylanases. CBM9 from Thermotoga maritima xylanase 10A is reported to have specificity for polysaccharide reducing ends. |
| pfam06452 | CBM9_1 | 2.03e-23 | 358 | 525 | 4 | 181 | Carbohydrate family 9 binding domain-like. CBM9_1 is a C-terminal domain on bacterial xylanase proteins, and it is tandemly repeated in a number of family-members. The CBM9 module binds to amorphous and crystalline cellulose and a range of soluble di- and monosaccharides as well as to cello- and xylo- oligomers of different degrees of polymerization. Comparison of the glucose and cellobiose complexes during crystallisation reveals surprising differences in binding of these two substrates by CBM9-2. Cellobiose was found to bind in a distinct orientation from glucose, while still maintaining optimal stacking and electrostatic interactions with the reducing end sugar. |
| cd00413 | Glyco_hydrolase_16 | 6.11e-23 | 82 | 319 | 1 | 210 | glycosyl hydrolase family 16. The O-Glycosyl hydrolases are a widespread group of enzymes that hydrolyse the glycosidic bond between two or more carbohydrates, or between a carbohydrate and a non-carbohydrate moiety. A glycosyl hydrolase classification system based on sequence similarity has led to the definition of more than 95 different families inlcuding glycosyl hydrolase family 16. Family 16 includes lichenase, xyloglucan endotransglycosylase (XET), beta-agarase, kappa-carrageenase, endo-beta-1,3-glucanase, endo-beta-1,3-1,4-glucanase, and endo-beta-galactosidase, all of which have a conserved jelly roll fold with a deep active site channel harboring the catalytic residues. |
| cd08023 | GH16_laminarinase_like | 8.20e-18 | 81 | 284 | 2 | 198 | Laminarinase, member of the glycosyl hydrolase family 16. Laminarinase, also known as glucan endo-1,3-beta-D-glucosidase, is a glycosyl hydrolase family 16 member that hydrolyzes 1,3-beta-D-glucosidic linkages in 1,3-beta-D-glucans such as laminarins, curdlans, paramylons, and pachymans, with very limited action on mixed-link (1,3-1,4-)-beta-D-glucans. |
CAZyme Hits help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End |
|---|---|---|---|---|---|
| QGG56400.1 | 6.30e-106 | 52 | 525 | 179 | 638 |
| AXT50244.1 | 5.12e-41 | 38 | 319 | 21 | 284 |
| CAL66698.1 | 5.26e-41 | 38 | 320 | 23 | 287 |
| AXT60531.1 | 2.01e-40 | 34 | 320 | 19 | 287 |
| AXT54175.1 | 5.65e-40 | 38 | 322 | 23 | 289 |
PDB Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| 4ATE_A | 1.36e-24 | 68 | 290 | 12 | 227 | Highresolution crystal structure of beta-porphyranase A from Zobellia galactanivorans [Zobellia galactanivorans] |
| 3ILF_A | 1.98e-22 | 68 | 290 | 14 | 229 | Crystalstructure of porphyranase A (PorA) in complex with neo-porphyrotetraose [Zobellia galactanivorans] |
| 3JUU_A | 1.50e-19 | 64 | 296 | 16 | 253 | Crystalstructure of porphyranase B (PorB) from Zobellia galactanivorans [Zobellia galactanivorans],3JUU_B Crystal structure of porphyranase B (PorB) from Zobellia galactanivorans [Zobellia galactanivorans] |
| 1I82_A | 3.34e-18 | 342 | 527 | 2 | 189 | Family9 Carbohydrate-Binding Module From Thermotoga Maritima Xylanase 10a With Cellobiose [Thermotoga maritima],1I8A_A Family 9 Carbohydrate-Binding Module From Thermotoga Maritima Xylanase 10a With Glucose [Thermotoga maritima],1I8U_A Family 9 Carbohydrate-Binding Module From Thermotoga Maritima Xylanase 10a [Thermotoga maritima] |
| 4AWD_A | 5.80e-18 | 73 | 337 | 47 | 309 | ChainA, BETA-PORPHYRANASE [Phocaeicola plebeius],4AWD_B Chain B, BETA-PORPHYRANASE [Phocaeicola plebeius] |
Swiss-Prot Hits download full data without filtering help
| Hit ID | E-Value | Query Start | Query End | Hit Start | Hit End | Description |
|---|---|---|---|---|---|---|
| D7GXG0 | 1.60e-22 | 68 | 290 | 23 | 238 | Beta-porphyranase A OS=Zobellia galactanivorans (strain DSM 12802 / CCUG 47099 / CIP 106680 / NCIMB 13871 / Dsij) OX=63186 GN=porA PE=1 SV=1 |
| D7GXG3 | 3.35e-21 | 73 | 298 | 28 | 255 | Beta-porphyranase E OS=Zobellia galactanivorans (strain DSM 12802 / CCUG 47099 / CIP 106680 / NCIMB 13871 / Dsij) OX=63186 GN=porE PE=3 SV=1 |
| D7GXF9 | 1.02e-18 | 64 | 296 | 29 | 266 | Beta-porphyranase B OS=Zobellia galactanivorans (strain DSM 12802 / CCUG 47099 / CIP 106680 / NCIMB 13871 / Dsij) OX=63186 GN=porB PE=1 SV=1 |
| O69230 | 1.15e-18 | 277 | 527 | 823 | 1085 | Endo-1,4-beta-xylanase C OS=Paenibacillus barcinonensis OX=198119 GN=xynC PE=1 SV=1 |
| P38535 | 1.45e-17 | 340 | 525 | 709 | 894 | Exoglucanase XynX OS=Acetivibrio thermocellus OX=1515 GN=xynX PE=3 SV=1 |
SignalP and Lipop Annotations help
This protein is predicted as SP
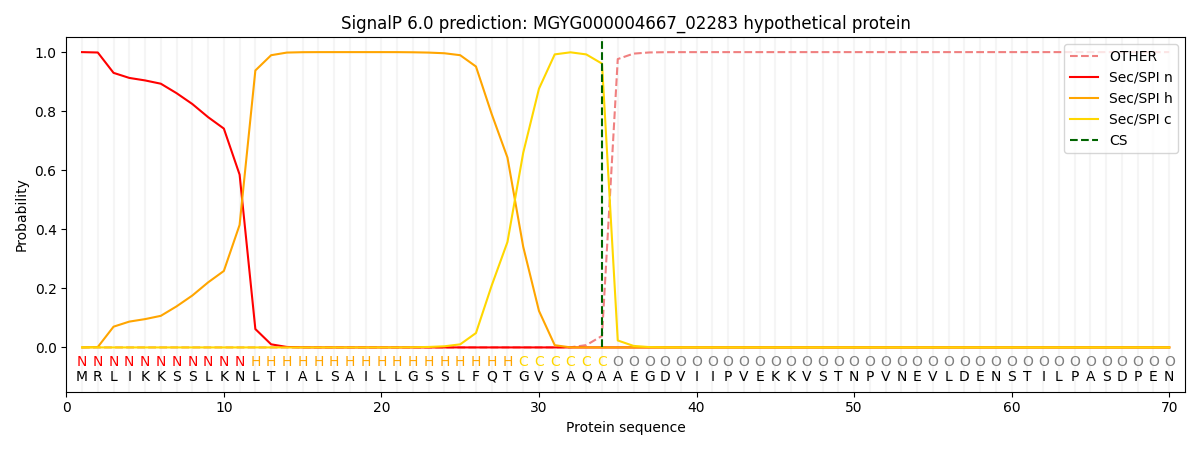
| Other | SP_Sec_SPI | LIPO_Sec_SPII | TAT_Tat_SPI | TATLIP_Sec_SPII | PILIN_Sec_SPIII |
|---|---|---|---|---|---|
| 0.000318 | 0.998942 | 0.000208 | 0.000202 | 0.000161 | 0.000141 |
